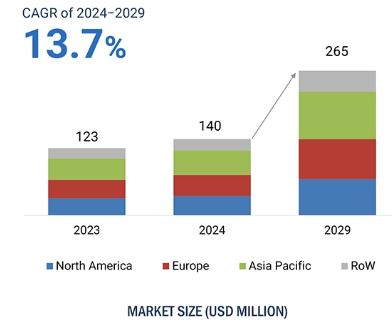
In recent years, Vitamin K2 has gained increasing attention across the nutrition and health industry. According to the latest market data, the global Vitamin K2 market is projected to grow from USD 140 million in 2024 to USD 265 million by 2029, with a compound annual growth rate (CAGR) of up to 13.7%.
As a key nutritional ingredient for supporting bone health and reducing the risk of arterial calcification, Vitamin K2 is often formulated in combination with Vitamin D3, calcium, magnesium, and other nutrients to create synergistic nutritional solutions. This combined formulation strategy has become a mainstream R&D trend in end products, with leading brands such as BY-HEALTH and Caltrate already launching related products.
However, studies have shown that during actual formulation processes, Vitamin K2 can undergo significant degradation when it comes into contact with minerals such as calcium and magnesium. This means that end products may fail to maintain the labeled content throughout their shelf life, potentially leading to formulation inefficacy, compliance risks, and brand reputation damage.
To address the degradation challenges of Vitamin K2 during shelf life, the industry has increasingly adopted encapsulation technologies. However, currently available encapsulated K2 products in the Chinese market often offer limited protective capability and still struggle to maintain long-term stability in calcium- and magnesium-rich systems.
To tackle this common industry challenge, RICHEN has developed Rivik2® Encapsulated Powder through dedicated research by its professional technical team — a Vitamin K2 ingredient featuring an exclusive microcapsule structural formulation.
How Does Rivik2® Achieve “Dual Protection”?
Rivik2® Encapsulated Powder adopts a microcapsule protective structure based on food-grade safe materials. Through targeted formulation design and spray-drying technology, a protective barrier is formed around Vitamin K2, effectively preventing direct contact with minerals such as calcium and magnesium and avoiding degradation.
This ensures that Vitamin K2 content remains stable throughout complex formulation systems and across the entire product shelf life, thereby safeguarding product efficacy.
Benefiting from this dual-protection system, Rivik2® demonstrates significantly improved stability in complex formulations, especially in products with extended shelf life and those containing calcium and magnesium. After a 3-month accelerated stability test, the retention rate of Vitamin K2 (MK-7) remained as high as 99.7% in formulations containing Vitamin D3, calcium carbonate, and magnesium oxide, highlighting its outstanding stability performance.
Three Core Advantages of Rivik2® Encapsulated Powder
Enhanced Stability (Processing & Storage Protection)
Compared with conventional Vitamin K2 powders and other encapsulated products, microscopic analysis shows that Rivik2® features more uniform particle size and more regular morphology, improving formulation homogeneity during processing.
Additionally, the microcapsule protective structure minimizes degradation caused by light exposure and calcium/magnesium systems, significantly improving Vitamin K2 content stability and reducing the risk of label claim non-compliance during shelf life — providing more reliable formulation compliance assurance for brands.
Improved Application Performance (Better Dispersibility)
Vitamin K2 is a fat-soluble vitamin that is naturally insoluble in water, which limits its application across various product formats. Through optimized structural design, Rivik2® Encapsulated Powder significantly improves the dispersibility of Vitamin K2, enabling easy incorporation into oral liquids, gummies, powdered milk formulas, and other delivery formats.
Analytical Method Support
RICHEN’s China National Accreditation Service for Conformity Assessment (CNAS)-accredited testing center has optimized the pre-treatment procedures for encapsulated Vitamin K2 based on the official testing methods announced by the National Health Commission of China, and completed full methodological validation to ensure scientific accuracy and reliability of test results.
RICHEN is able to provide customers with the complete validated testing method, offering strong support for quality control and compliance management.
RICHEN Nutrition has completed comprehensive stability validation of Rivik2® across various formulation systems, ensuring its superior performance in end-product applications.